| 生物活性 | |||
|---|---|---|---|
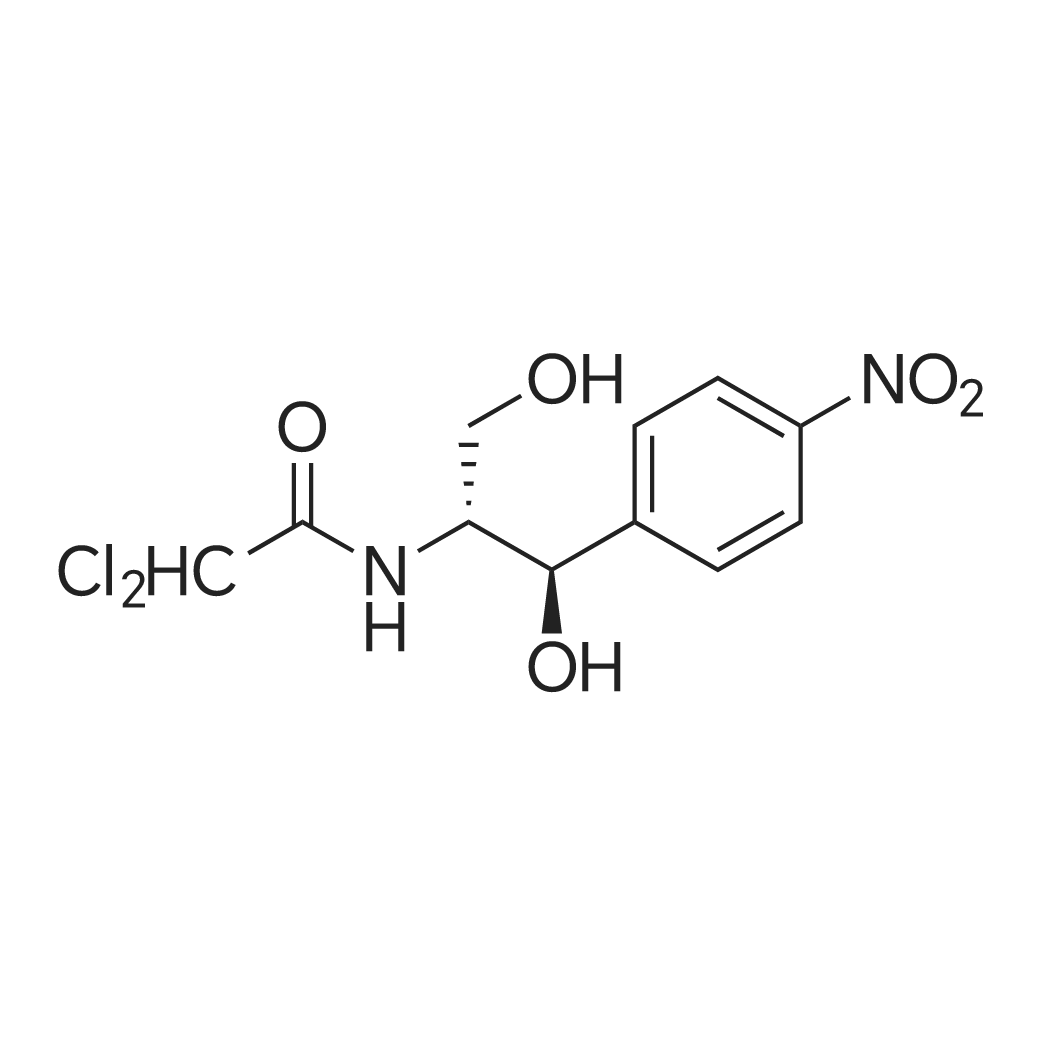
| 描述 | Chloramphenicol is a unique antibiotic. The kinetics and efficacy of the oral and intravenous preparations are comparable. Chloramphenicol is usually bacteriostatic but is bactericidal against Haemophilus influenzae, Streptococcus pneumoniae, and Neisseria meningitidis, and chloramphenicol's clinical efficacy against these meningeal pathogens is well established[3]. The common, dose-dependent, reversible bone marrow suppression from chloramphenicol is a consequence of mitochondrial injury. The greater erythroid susceptibility to chloramphenicol appears to be a function of the endogenous mitochondrial amino acid pools[4]. Depletion in compounds acting as cellular antioxidants, such as glutathione and vitamin E, may conceivably increase the vulnerability of an individual to chloramphenicol toxicity, while supplementation with an antioxidant might protect against it[5]. Three preparations of chloramphenicol are most commonly used in clinical practice: a crystalline powder for oral administration, a palmitate ester for oral administration as a suspension, and a succinate ester for parenteral administration. Both esters are inactive, requiring hydrolysis to chloramphenicol for anti-bacterial activity[6]. | ||
| 实验方案 | |||
|---|---|---|---|
| 1mg | 5mg | 10mg | |
|
1 mM 5 mM 10 mM |
3.09mL 0.62mL 0.31mL |
15.47mL 3.09mL 1.55mL |
30.95mL 6.19mL 3.09mL |
| 参考文献 |
|---|
|
[4]Yunis AA. Chloramphenicol toxicity: 25 years of research. Am J Med. 1989;87(3N):44N–48N |